A leading life sciences manufacturer faced a critical obsolescence risk on one of its highest-volume packaging lines, which operated with over 120 servo axes controlled by the Rockwell 1394 motion platform. With the platform discontinued and spare parts nearly impossible to source, any failure could have resulted in weeks of unplanned downtime and jeopardized compliance in an FDA-regulated environment. Joltek was engaged to lead the full retrofit of the system, migrating to the Rockwell Kinetix 6200 platform while upgrading controller firmware and validating all motion sequences. Through three months of preparation and a tightly managed two-week shutdown, the migration was completed on schedule, fully validated, and returned to production with improved reliability and long-term supportability.
The client is a global life sciences manufacturer operating under strict FDA regulatory oversight. Every production line is subject to Good Manufacturing Practices (GMP) and must be validated through Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Any system failure not only risks significant production losses but also carries compliance and documentation consequences.
The packaging line in focus is one of the facility’s highest-volume assets, responsible for critical throughput in a market with high product demand. The line was originally equipped with over 120 servo axes running on the Rockwell 1394 motion platform, controlled through a SERCOS fiber-optic ring that required precise termination and continuity. The servo drives interfaced with ControlLogix 1756-L6x controllers and operated under legacy HMI and SCADA systems.
This platform, while once state-of-the-art, had reached end-of-life status. Spare parts were scarce, technical support was minimal, and the risk of a single drive or feedback device failure was escalating rapidly. A catastrophic failure could have resulted in weeks of downtime, costing millions of dollars in lost output and compromising FDA compliance.
The stakes for the client included:

By proactively addressing these risks, the client sought to modernize its motion platform with minimal disruption while establishing a path forward for long-term reliability.
The existing motion control system, built on the Rockwell 1394 platform, exposed the facility to significant risks across multiple dimensions.
Technical Risks
Operational Risks
Compliance Risks
Business Risks
Joltek defined clear objectives and success criteria to ensure a successful migration while protecting business continuity.
Migration Objectives
Performance Objectives
Compliance Objectives
Before any retrofit could begin, it was necessary to establish a clear picture of the existing system and the risks associated with changing it. The first step was a full asset inventory of the motion platform, which included over one hundred and twenty axes of motion, the 1394 drives, feedback devices, cabling, and associated power infrastructure. Every servo motor, encoder, and connection had to be identified and documented, since even a single oversight could have caused significant delays once the shutdown began.
The controls architecture was reviewed in detail. The system was running on a SERCOS fiber optic ring that connected the 1394 platform to the ControlLogix 1756 L6x controllers. This topology required precise termination and clean communication between nodes, and the age of the hardware made it important to validate continuity and prepare for potential issues during the cutover. The controllers themselves were operating on firmware that was behind current standards, which meant that upgrades would be necessary to maintain compatibility with the new Kinetix 6200 drives.
Motion logic and application software were also baselined. Axis configurations, homing routines, cam profiles, torque limits, and interlocks had to be extracted and mapped so that the migration could be executed with confidence. Golden recipes for motion performance were captured, including cycle times, acceleration curves, and following error tolerances. This data would become the reference point for qualification testing once the new drives were installed.
Power and thermal considerations were assessed, with particular focus on regeneration handling. The legacy platform used both internal and external shunt resistors to manage energy during deceleration, and it was essential to confirm that equivalent or greater capacity would be in place after the migration. Failure to properly size shunt capacity can damage hardware or cause nuisance trips that compromise reliability.
Feedback devices were another critical element of the baseline. The 1394 system supported a range of feedback types, including resolvers, TTL, sine cosine, and Hiperface. Each axis needed to be checked for compatibility and migration planning, since differences in connectors and pinouts could become a hidden source of rework if not addressed in advance.
Finally, documentation of compliance requirements was part of the baseline effort. This included identifying the scope of validation that would be required under FDA guidelines and mapping each system change to the appropriate IQ, OQ, and PQ activities. By aligning these requirements with the technical baseline, the team was able to build a project plan that accounted for both engineering and regulatory expectations.
The migration strategy had to balance technical feasibility, regulatory compliance, and strict time constraints. With over one hundred and twenty axes involved, even small errors in planning could have compounded into significant delays. The approach taken was to focus on minimizing disruption, preserving compatibility where possible, and applying best practices that reduced the likelihood of surprises during the shutdown window.
One of the first design decisions was to remain on the SERCOS communication protocol by selecting the Kinetix 6200 platform. Although the Kinetix 6500 offered EtherNet/IP motion, shifting to a different protocol would have expanded the scope of changes well beyond what the shutdown allowed. By maintaining SERCOS, the project preserved compatibility with the existing ControlLogix 1756 L6x controllers while still enabling a path to future modernization. This choice highlighted an important best practice: avoid unnecessary scope creep during obsolescence projects and instead establish a phased roadmap that can be executed over time.
Safety functionality was another major consideration. The legacy platform had no integrated safety features, which created an opportunity to introduce modern standards during the migration. The Kinetix 6200 platform supported Safe Torque Off and Safe Speed Monitoring, both of which were evaluated against the facility’s risk assessments and performance level requirements. The best practice here was to align safety upgrades with the broader modernization effort, ensuring that changes deliver not only compliance but also long-term operator and maintenance benefits.
The electrical and power design required careful planning. The new drive modules had to be sized for proper load distribution, thermal management, and regeneration handling. The 1394 platform had relied on both internal and external shunt resistors, so the design team validated that the new system had equivalent or greater regeneration capacity. In high axis-count systems, overlooking shunt sizing is a common pitfall that leads to nuisance trips and reliability issues, so confirming this early in the design process was essential.
Feedback device compatibility was one of the more complex challenges. The 1394 platform supported multiple feedback types including resolver, sine cosine, TTL, and Hiperface. Each axis was reviewed for compatibility, and where necessary, connector kits and wiring adapters were planned in advance. The best practice in this area is to document every feedback device and cable at the start of the project, since encoder incompatibility is one of the most common causes of delays during a large retrofit.
Software planning was another area where attention to detail mattered. Axis configurations, homing sequences, cam profiles, and interlocks were translated to the Logix environment used by the Kinetix 6200. This process also included setting new motion parameters and retuning axes once the drives were installed. A best practice followed here was to create a parameter map that compared old values to new values, ensuring that no settings were overlooked during the transition.
Finally, compliance and validation were integrated directly into the migration strategy rather than treated as a separate step. IQ, OQ, and PQ activities were mapped to each stage of the project so that testing and documentation could be performed in parallel with technical work. This approach not only accelerated the validation timeline but also ensured that quality and regulatory expectations were met without creating delays after startup.

To summarize, the best practices applied during the strategy phase included:
By applying these practices, the project was set up for success long before the first drive was replaced, which ultimately allowed the shutdown execution to proceed on time and without major surprises.
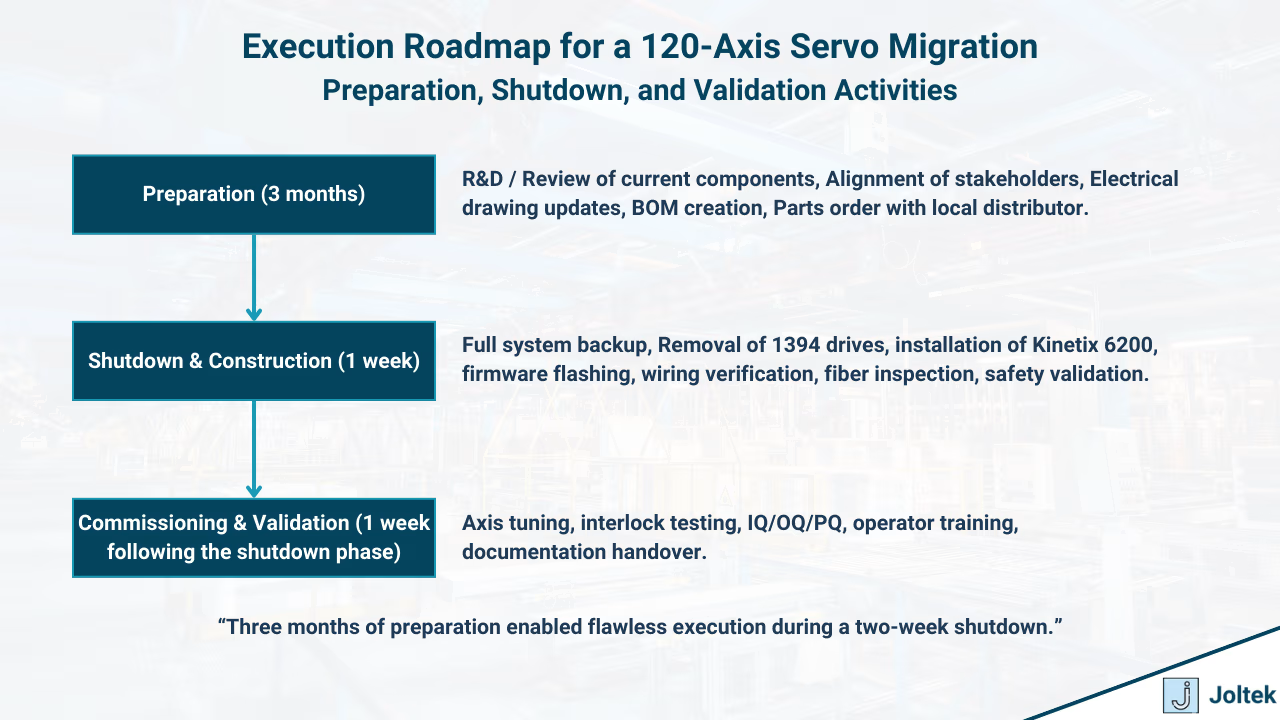
The migration was structured around three phases: preparation, the two week shutdown, and post installation validation. Preparation lasted three months and was the foundation of success. Every axis was mapped, feedback devices were documented, and golden data was captured from the existing system to serve as a performance baseline. Bench testing was completed to confirm firmware compatibility between the ControlLogix controllers and the Kinetix 6200 drives. The best practice here was clear: preparation must be treated as execution, since every risk addressed in advance prevents delays during the shutdown.
The two week cutover was managed with a detailed day by day playbook. Teams worked in parallel to remove the 1394 drives, install the new rails, flash firmware, and validate wiring and encoder connections. The SERCOS ring was inspected and cleaned to prevent communication issues. Strict checklists ensured that no step was skipped, which is a best practice in large retrofits where a single missed verification can cost days of recovery.
Commissioning and validation followed immediately after installation. Each axis was tuned and tested against the original golden data. Safety functions, interlocks, and fault handling were verified before full speed trials. Validation was fully aligned with FDA requirements and followed IQ, OQ, and PQ protocols. Training, updated documentation, and a thirty day hypercare period were provided to support long term success.
By combining rigorous preparation, disciplined execution, and integrated validation, the project achieved its objectives within the planned shutdown and returned to production without deviation.
The migration was completed within the planned two week shutdown window and the line was released back to production on schedule. More importantly, the startup was achieved without a single deviation, which is a rare outcome for a project of this scale. By addressing obsolescence proactively, the facility avoided the risk of a sudden failure that could have caused weeks of unplanned downtime and millions in lost output.
The modernized motion platform delivered improved reliability across all one hundred and twenty axes. Servo related faults were reduced significantly, and the system’s enhanced diagnostic capabilities made troubleshooting faster and more precise. Integrated safety functionality provided additional protection and aligned the line with current standards, giving operators greater confidence in daily operation.
Validation was completed successfully under FDA and GMP requirements, with all IQ, OQ, and PQ protocols approved without rework. The project not only delivered technical success but also met every compliance expectation. From a financial perspective, the avoided downtime risk and reduction in unplanned maintenance translated into a payback period of less than eighteen months.
In the end, the retrofit did more than replace an obsolete platform. It improved the performance and reliability of a critical production line, strengthened compliance posture, and provided a clear roadmap for future modernization.

Large retrofits like this one always surface lessons that extend well beyond the technical scope of the project. One of the most important takeaways was the value of detailed encoder and feedback documentation. In many migrations, hidden compatibility issues only become apparent once the new drives are powered up, which can cause days of troubleshooting. By cataloging every feedback device in advance and preparing connector kits ahead of time, the team eliminated what is often the single largest source of delay in servo migrations.
Another lesson was the importance of maintaining the integrity of the SERCOS communication ring. Even with proper hardware, contamination on a fiber connection or a poorly terminated segment can bring an entire line offline. Taking the time to inspect and clean every fiber during installation was a simple step that ensured communication was stable from startup onward.
The project also reinforced the need to approach axis tuning with a balance of speed and rigor. Attempting to retune one hundred and twenty axes without a structured plan could have easily extended the shutdown timeline. Instead, the team focused on representative groups of axes, verified performance against golden data, and then scaled those parameters across similar motion groups. This method delivered both accuracy and efficiency.
Finally, the integration of validation into the technical plan was essential. Treating compliance as a separate effort would have slowed startup and created unnecessary friction between engineering and quality. By mapping every test protocol directly to commissioning tasks, validation became a natural part of the workflow rather than a hurdle to overcome at the end.
The broader lesson is that modernization projects succeed when technical execution, operational readiness, and compliance requirements are treated as inseparable parts of the same strategy. This alignment was the reason the migration finished on time, within scope, and without deviations.